ACS Sens. 2021, 6, 11, 3912–3932,Full text
Shanshan Lin【林珊珊】, Ziyi Cheng【程子译】, Qifu Li【李其富】*, Rui Wang【王锐】*, and Fabiao Yu【于法标】*
Publication Date:November 2, 2021 https://doi.org/10.1021/acssensors.1c01858
Copyright © 2021 American Chemical Society
Abstract
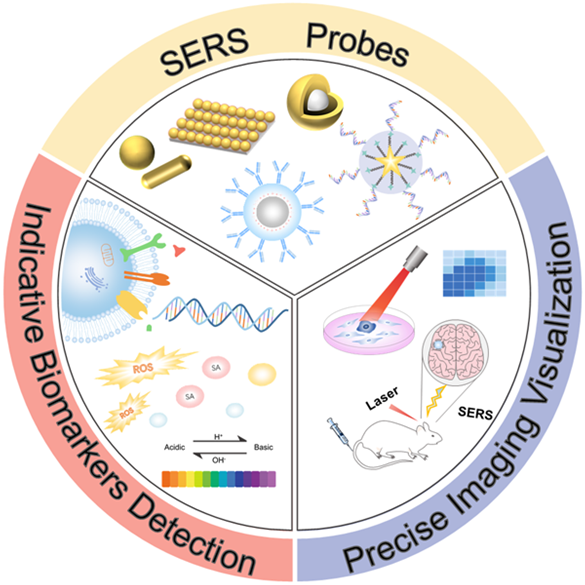
Early specific detection through indicative biomarkers and precise visualization of lesion sites are urgent requirements for clinical disease diagnosis. However, current detection and optical imaging methods are insufficient for these demands. Molecular imaging technologies are being intensely studied for reliable medical diagnosis. In the past several decades, molecular imaging with surface-enhanced Raman scattering (SERS) has significant advances from analytical chemistry to medical science. SERS is the inelastic scattering generated from the interaction between photons and substances, presenting molecular structure information. The outstanding SERS virtues of high sensitivity, high specificity, and resistance to biointerference are highly advantageous for biomarker detection in a complex biological matrix. In this work, we review recent progress on the applications of SERS imaging in clinical diagnostics. With the assistance of SERS imaging, the detection of disease-related proteins, nucleic acids, small molecules, and pH of the cellular microenvironment can be implemented for adjuvant medical diagnosis. Moreover, multimodal imaging integrates the high penetration and high speed of other imaging modalities and imaging precision of SERS imaging, resulting in final complete and accurate imaging outcomes and exhibiting robust potential in the discrimination of pathological tissues and surgical navigation. As a promising molecular imaging technology, SERS imaging has achieved remarkable performance in clinical diagnostics and the biomedical realm. It is expected that this review will provide insights for further development of SERS imaging and promote the rapid progress and successful translation of advanced molecular imaging with clinical diagnostics.
Figure 1. (A) Nanotube arrays (NTAs). SEM (ⅰ, ⅱ) and TEM (ⅲ) images of TiO2 NTAs. SEM (ⅳ, ⅴ) images of TiO2/Au NTAs. Insert in ⅳ: EDX characterization of TiO2/Au NTAs. Raman spectra (ⅵ) of pMBA from 15 random positions on TiO2/Au NTAs. Adapted with permission from Ref. 24. Copyright 2018 Elsevier. (B) Nanocages (AgNCs). Typical TEM images of AgNCs (ⅰ), AuNCs (ⅱ), AuNC/SiO2 NPs (ⅲ), and AuNC/SiO2/Apt (ⅳ). Insets: HRTEM images of AgNCs (ⅰ) and AuNCs (ⅱ). Scale bars: 20 (insets) and 50 nm. Adapted with permission from Ref. 26. Copyright 2019 American Chemical Society. (C) Nanomushrooms. Up) TEM images of Au-Ag nanomushrooms using AuNP seeds with the diameter of 50 nm. Down) SEM images of sandwich MMP complexes. Left: In the presence of target DNA (10 nM), a large number of 100 nm Au-Ag nanomushrooms were coupled with the MMPs due to DNA hybridization. Right: The blank control experiment in which no DNA target was added. Adapted with permission from Ref. 28. Copyright 2017 American Chemical Society. (D) Nanoflowers (AuNFs). ⅰ, TEM image of AuNFs; ⅱ, TEM image of single AuNF and its interior nanogap; ⅲ, UV-vis spectrums of AuNPs and AuNFs; ⅳ, finite-difference time-domain simulation of electromagnetic field distribution of AuNFs. Adapted with permission from Ref. 27. Copyright 2019 John Wiley and Sons. (E) Nanocorals. Schematic of the nanofabrication process of WTNC SERS substrates (ⅰ-ⅲ). A cross-sectional SEM image (ⅳ) and a top-view SEM image (ⅴ) of a wafer-scale polymer-based transparent nanocoral (WTNC) substrate, produced by 300 s RIE of highly crosslinked SU-8 2035 followed by e-beam evaporation of 200 nm thick gold. ⅵ, a photograph of a 4 in. WTNC SERS substrate, taken from its backside. The grid is ascribed to the use of a shadow mask during e-beam evaporation of gold, leaving room for subsequent processes such as dicing to obtain SERS chips and microfluidic integration. ⅶ, two SERS spectra of BPE, obtained respectively from the front and the backside of a WTNC substrate produced with optimal parameters, averaged over the same area containing 25 points. Adapted with permission from Ref. 29. Copyright 2019 John Wiley and Sons. (F) Core-Satellite Nanostructures. ⅰ, ⅱ, TEM images of Au NDs and Au NPs-Au NDs CS (mir-1246:0.5 μM), respectively. ⅲ, DLS intensity and ⅳ, UV-vis absorption spectra of Au NPs, Au NRs, Au NDs, and Au NPs-Au NDs CS. Adapted with permission from Ref. 91. Copyright 2018 American Chemical Society.
Figure 2. (A, B) Schematic for the Cleave-and-Bind Mechanism for the logical multiplex detection of MMPs on nanopillar chip and bright-field image of nanopillar chips and SERS mapping images. (Scale bar = 10 μm). Adapted with permission from Ref. 64. Copyright 2017 American Chemical Society. (C, D) Schematic illustration for the multiplexed detection of Alzheimer’s biomarker Aβ40 and Aβ42 over Aβ antibody-conjugated AgNGS nanoprobes in a sandwich immunoassay and detection of Alzheimer biomarker Aβ40 and Aβ42 over AgNGS nanoprobes. Raman mapping images of CAb-MB dropped on a glass array after incubation with AgNGS[4-FBT]–Aβ40 and AgNGS[4-BBT]–Aβ42, respectively. Adapted with permission from Ref. 67. Copyright 2019 John Wiley and Sons.
Figure 3. (A) ⅰ, Au@pNIPAM SERRS tags indicating their Raman codification and targeting entities. ⅱ, Schematic representation of the SERRS immunophenotype detection of A431 and 3T3 2.2 cells. Adapted with permission from Ref. 70. Copyright 2015 John Wiley and Sons. (B) ⅰ, schematic illustration of pattern recognition and discrimination of cancer cell lines with multiplexed GIAN-encoders. ⅱ, SERS images of cancer cells, scale bar, 10 μm. G100, G050 and G000 conjugated with DSPE-PEG-linked aptamer AS1411, S1.6 and SYL3C, respectively. ⅲ, SERS barcodes of HepG2 and A549 cell lines and ⅳ, statistics of normalized SERS signals. Adapted with permission from Ref. 71. Copyright 2018 Royal Society of Chemistry.
Figure 4. (A) Whole abdomen imaging of representative control (left) and tumor-bearing (right) mice. Bioluminescence (BLI) signal is shown in the top row. The DCLS maps of both targeted (2nd row) and nontargeted (3rd row) show a nonspecific distribution of both probes throughout the peritoneal cavity. TAS3RS (4th row) shows no positive regions in the control (left) and a strong correlation to BLI in tumorbearing mice (right). Alternatively the TAS3RS map can be visualized in a simplified manner for surgical guidance (bottom row), showing only regions with positive ratios in red. Reference standard solutions in Eppendorf vials were placed on the imaged field of view, with (1) indicating the vial containing αFR-NPs and (2) the vial containing nt-NPs. Adapted with permission from Ref. 74. Copyright 2017 American Chemical Society. (B, C) Schematic illustration of DNA Alkyne-functionalized alloyed Au/Ag nanoparticle-based SERS nanosensor for ratiometric detection of endonuclease and SERS imaging of nanoprobe-loaded HeLa cells, before (up) and after (down) treatment with 2-phenethyl isothiocyanate (PEITC). SERS spectra and ratiometric peak I1983/ I2215 were obtained from points (a-c, d-f) denoted in the Raman ratiometric images in the same row. I1983/I2215 Raman ratiometric images are displayed in pseudocolor. Scale bar = 10 μm. Adapted with permission from Ref. 77. Copyright 2018 American Chemical Society.
Figure 5. (A) Schematic illustration of the Turn-On Probe for detecting GGT based on PEF and SERS Techniques. Adapted with permission from Ref. 79. Copyright 2019 American Chemical Society. (B) Schematic Representation of the fabrication routes of Au NPs-Au NDs CS and Au NPs-Au NDs Core–Satellite (CS) SERS sensors for miRNA detection and imaging in living Cells. Adapted with permission from Ref. 91. Copyright 2018 American Chemical Society.
Figure 6. (A, B) Working principle of the present single SERS nanoprobe for imaging and biosensing of ClO– and GSH. (ⅰ1–ⅰ3) Bright-field, dark-field, and overlay images of RAW264.7 cells after 4 h incubation with AuF/MP nanoprobes. (ⅱ1–ⅱ3 and ⅲ1–ⅲ3) SERS imaging of AuF/MP nanoprobes in RAW264.7 cells: (ⅱ1 and ⅲ1) in the absence of ClO– and GSH; (ⅱ2 and ⅲ2) induced by PMA; (ⅱ3, ⅲ3) after incubation with GSH for 30 min followed by stimulation by PMA. (ⅱ represents I635/I822 and ⅲ represents I1077/I822). Adapted with permission from Ref. 106. Copyright 2016 American Chemical Society.

Figure 7. (A) ⅰ, working principle of the PB-caged SERS probe. ⅱ, PB as a background-free internal standard for profiling the distribution of PB-caged 4-MPy–AuNP pH probes in live cells. Bright-field and Raman mapping of the 1097 and 2138 cm–1 channels and a merged image of the HeLa cell incubated with the proposed probes. The images were scanned at intervals of 1 μm with 633 nm excitation and an exposure time of 0.1 s. Scale bar = 20 μm. ⅲ, intracellular pH quantification induced by starvation. pH imaging of the probe-treated HeLa cells that were incubated in nutrient-rich culture medium (normal culture condition), nutrient-deficient culture medium (starvation condition), and in the starvation condition with treatment by chloroquine (100 μM). Scale bars = 20 μm. Adapted with permission from Ref. 117. Copyright 2020 American Chemical Society. (B) ⅰ, schematic illustration of the stepwise synthesis of PA-engineered pH probes (AuNS-4MBA-PA). ⅱ, reconstruction of 3D SERS imaging at time points 2 h and 8 h on the same cell and corresponding Raman spectra. Adapted with permission from Ref. 118. Copyright 2020 American Chemical Society.
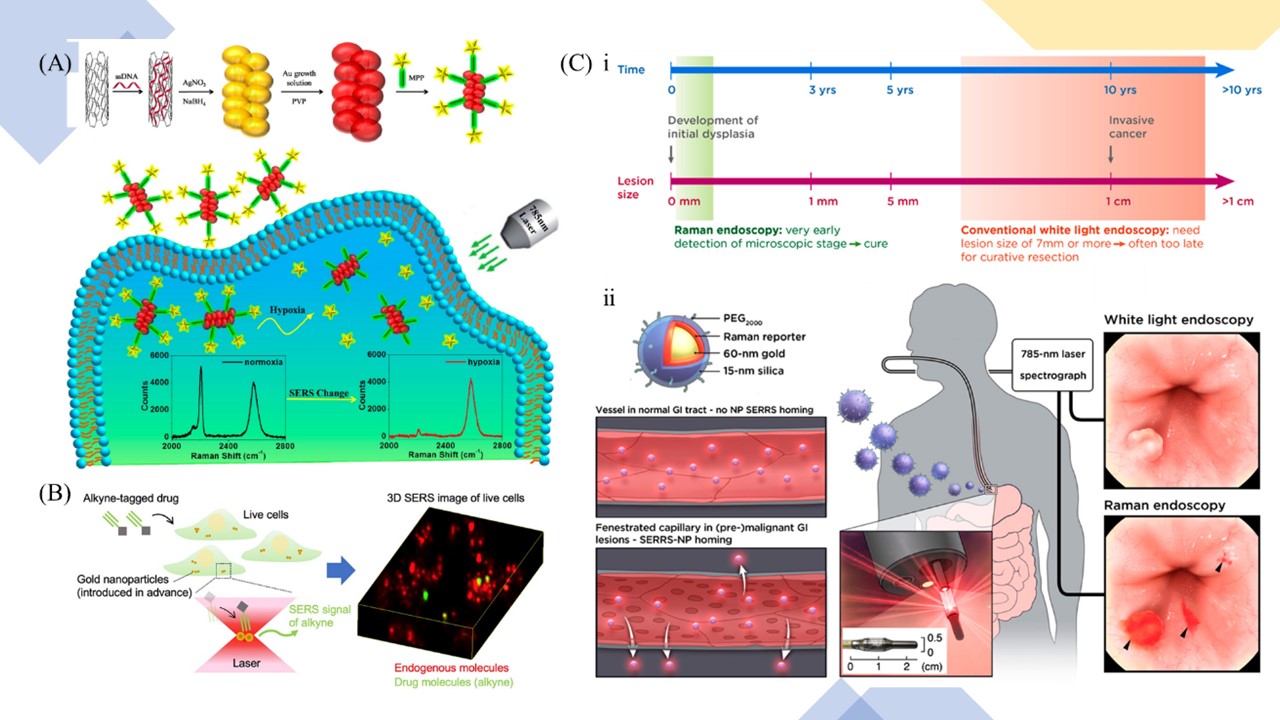
Figure 8. (A) The preparation of the SWCNT/Ag/AuNPs/MPP conjugate-based SERS nanoprobe and the sensing mechanism of hypoxia. Adapted with permission from Ref. 125. Copyright 2019 American Chemical Society. (B) Schematic diagram of 3D SERS imaging of uptake distribution information of a small molecule drug labeled with alkyne in living cells. Adapted with permission from Ref. 128. Copyright 2020 American Chemical Society. (C) Concept of contrast-enhanced Raman imaging of gastrointestinal lesions. ⅰ, comparison of lesion detectability between SERRS-NP-enhanced Raman endoscopy and conventional white-light endoscopy currently used in the clinic. ⅱ, rendered cross-section diagram of the SERRS-NPs used, envisioned clinical use: intravenously injected SERRS-NPs specifically accumulate in (pre-) malignant lesions and illustration of the acquired imaging data. Adapted with permission from Ref. 131. Copyright 2019 American Chemical Society.
Figure 9. (A) Guiding brain-tumor surgery by acid-responsive gold nanoprobes. Up) The cleavage of the PEG coating in physiological acidity triggers aggregation between Au–AZ and Au–AK via “click” cycloaddition reactions. Down) Due to the LPR1-mediated, bidirectional, BBB-traversing strategy, while the bulky AuNS aggregates are continuously trapped in tumor acidic interstitium, the intact nanoprobes in the normal brain tissue can be transported back into the blood stream, which increases the sensitivity for the brain-tumor margins. Adapted with permission from Ref. 137. Copyright 2017 John Wiley and Sons. (B, C) Schematic design principle of FRNPs and the optimized FRNPs are used for dual-mode cancer imaging and photothermal therapy and in vivo cancer imaging and photothermal therapy using OFRNPs. Adapted with permission from Ref. 138. Copyright 2019 Springer Nature.
Summary and outlook
SERS is a robust analytical and imaging tool that provides specific molecular vibration and rotation information with ultra-sensitivity and strong multiplexing capability. Decades of practical explorations allow SERS to be an increasingly mature technology that can be applied to many fields such as biology, chemistry, and agriculture. In this review, we elaborated on the extensive applications in the biological and biomedical field and focused on the detectability of SERS imaging in clinical diagnostics. With its unparalleled advantages, SERS imaging can realize the localization detection and real-time in vivo or in vitro imaging of targeted proteins, nucleic acids, pH, and other biological molecules. Various pathophysiological processes can be reflected by SERS imaging as well. Compared with basic SERS spectroscopy, SERS imaging can be used to monitor analytes more intuitively and visually. The combination of SERS imaging and endoscope to identify pathological areas is a massive advancement in transforming SERS into clinical practice. Remarkably, the multifunction platform integrated several optical technologies like MRI, PA imaging, fluorescence imaging, SERS imaging, et al., was effectively applied to preoperative and intraoperative image-guided surgical treatment with full advantages of integration elements. However, there are still some diagnostic practices we have not reviewed, such as lipid molecules and pathogen detection, which also prove the wide applicability of SERS imaging.139-141
For the development and application of SERS imaging in medical diagnostics in the future, a host of opportunities and challenges coexist, and the following points must be paid more attention to. (1) Although SERS imaging has been widely used in bioresearch and diagnostics, the limited resolution and imaging speed cannot be overlooked. For one thing, by adjusting SERS substrates and optimizing Raman reporters, signal amplification will be stimulated so as to reduce the acquisition time for SERS imaging in some extent.142,143 For another, considering that the current Raman microscope cannot meet the requirement of high-speed and high-resolution imaging, how to develop a suitable technique innovatively to implement a more formidable measurement function is a tremendous challenge for basic research and technical personnel. The super-solution SERS imaging for monitoring integrin receptors in cells was realized by developing a home-built wide-field Raman microscope, integrating an upright Olympus BX microscope with a CMOS camera.144 The study achieved a positioning precision of ≤ 6 nm and images were recorded at 100 ms per frame, which presented a promising experimental attempt, inspiring more new ideas and innovations. (2) The issue of improving penetration depth and reducing damages to biological samples is indeed of great concern in SERS imaging for the actual clinical diagnosis needs. SERS imaging of the NIR region, especially in the second NIR window, needs to be further investigated. More flexible substrates and reporters should be exploited to coordinate with NIR laser of 785 nm or even 808 nm to inspire maximum effectiveness. (3) For clinical laboratory diagnosis, there is a general trend of high-speed and high-throughput testing. Presently, many SERS-based microfluidic testing chips have been constructed.145,146 SERS-based microfluidics are promising to be an innovative diagnostic practice since it can realize high-throughput testing with a small amount of sample and labor, and is user-friendly, especially for point of care testing. We anticipated that SERS microfluidic would be a hot topic of research continuously. (4) The integration of diagnosis and therapy is another requirement of clinical practice for SERS development.147 SERS-based platform performs sensitive and specific identification and diagnosis, and then the targeting SERS probes produce photothermal or photodynamic effects under laser radiation or respond to the microenvironment to activate other cascade therapy plans. There is still huge room for development in the nano theranostics platform combing more advanced treatment modes. Monitoring the efficacy of therapeutic strategy is another biomedical application that can be extended.148 (5) In order to promote the applications of SERS probes in biological environment, especially for clinical translation, more factors need to be considered. Nanoprobes with more favorable biocompatibility are conducive to better biological applications and achieve a more controllable outcome of probes in the living body. Moreover, complicated environment in the organism may have additional and unpredictable effects on the detection of SERS probes, which is still worthy of further exploration. With the joint efforts of scientists and clinicians, we envision that more suitable SERS imaging probes and application platforms are bound to be developed for medical diagnosis and biomedical translation.