组会讲课人员:王振凯
Quantifying lysosomal glycosidase activity within cells using bis-acetal substrates
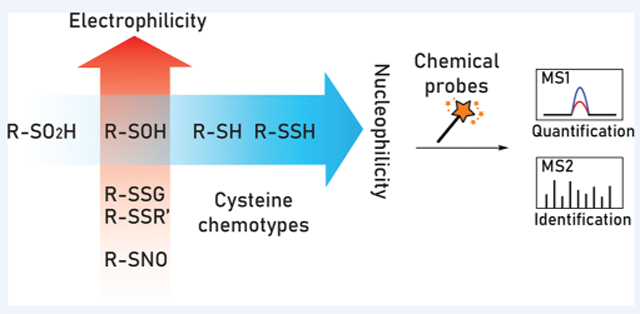
半胱氨酸氧化位点特异性蛋白质组学分析的基于活性的传感
主讲人:王振凯
Acc Chem Res,2020 Jan 21;53(1): 20-31. https://doi: 10.1021/acs.accounts.9b00562
Abstract:
Oxidative post-translational modifications (OxiPTMs) of cysteine residues are the molecular foundation of thiol-based redox regulation that modulates physiological events such as cell proliferation, differentiation, and migration and, when dysregulated, can lead to biomolecule damage and cell death. Common OxiPTMs of cysteine thiols (-SH) include reversible modifications such as S-sulfenylation (-SOH), S-glutathionylation (-SSG), disulfide formation (-SSR), S-nitrosylation (-SNO), and S-sulfhydration (-SSH) as well as more biologically stable modifications like S-sulfinylation (-SO2H) and S-sulfonylation (-SO3H). In the past decade, our laboratory has developed first-in-class chemistry-based tools and proteomic methods to advance the field of thiol-based redox biology and oxidative stress. In this Account, we take the reader through the historical aspects of probe development and application in our laboratory, highlighting key advances in our understanding of sulfur chemistry, in the test tube and in living systems. Offering superior resolution, throughput, accuracy, and reproducibility, mass spectrometry (MS)-based proteomics coupled to chemoselective “activity-based” small-molecule probes is the most rigorous technique for global mapping of cysteine OxiPTMs. Herein, we describe the evolution of this field from indirect detection to state-of-the-art site-centric quantitative chemoproteomic approaches that enable mapping of physiological and pathological changes in cysteine oxidation. These methods enable protein and site-level identification, mechanistic studies, mapping fold-changes, and modification stoichiometry. In particular, this Account focuses on activity-based methods for profiling S-sulfenylation, S-sulfinylation, and S-sulfhydration with an eye toward new reactions and methodologies developed in our group as well as their applications that have shed new light on fundamental processes of redox biology. Among several classes of sulfenic acid probes, dimedone-based C-nucleophiles possess superior chemical selectivity and compatibility with tandem MS. Cell-permeable dimedone derivatives with a bioconjugation handle are capable of detecting of S-sulfenylation in living cells. In-depth screening of a C-nucleophile library has yielded several entities with significantly enhanced reactivity over dimedone while maintaining selectivity, and reversible linear C-nucleophiles that enable controlled target release. C-Nucleophiles have also been implemented in tag-switch methods to detect S-sulfhydration. Most recently, activity-based detection of protein S-sulfinylation with electrophilic nitrogen species (ENS), such as C-nitroso compounds and electron deficient diazines, offers significant advantages in simplicity-of-use and target specificity compared to label-free methods. When feasible, the rich information provided by site-centric quantitative proteomics should not be tainted by oxidation artifacts from cell lysis. Therefore, chemoselective probes that function in a native environment with low cytotoxicity, good cell-permeability, and competitive kinetics are desired in modern redox chemoproteomics approaches. As our understanding of sulfur chemistry and redox signaling evolves, newly discovered cysteine OxiPTMs in microorganisms, plants, cells, tissues, and disease models should innovatively promote mechanistic and therapeutic research.
摘要:
半胱氨酸残基的氧化翻译后修饰 (OxiPTMs) 是基于硫醇的氧化还原调节的分子基础,可调节细胞增殖、分化和迁移等生理事件,当失调时,可导致生物分子损伤和细胞死亡。常见的半胱氨酸硫醇 (─SH) 的 OxiPTM 包括可逆修饰,例如 S-亚磺酰化 (─SOH)、S-谷胱甘肽化 (─SSG)、二硫键形成 (─SSR)、S-亚硝基化 (─SNO) 和 S-硫化( ─SSH) 以及更生物稳定的修饰,如 S-亚磺酰化 (─SO 2 H) 和 S-磺酰化 (─SO 3H)。在过去的十年中,我们的实验室开发了一流的基于化学的工具和蛋白质组学方法,以推进基于硫醇的氧化还原生物学和氧化应激领域。在这篇文章中,我们将带读者了解我们实验室中探针开发和应用的历史方面,重点介绍我们在了解硫化学、试管和生命系统方面取得的重要进展。
基于质谱 (MS) 的蛋白质组学与化学选择性“基于活性”的小分子探针相结合,提供卓越的分辨率、通量、准确度和重现性,是半胱氨酸 OxiPTM 全局映射最严格的技术。在这里,我们描述了该领域从间接检测到最先进的以位点为中心的定量化学蛋白质组学方法的演变,这些方法能够绘制半胱氨酸氧化的生理和病理变化图。这些方法可以实现蛋白质和位点级别的识别、机制研究、映射倍数变化和修饰化学计量。特别是,该帐户侧重于分析 S-亚磺酰化、S-亚磺酰化、和 S-硫化,着眼于我们小组开发的新反应和方法以及它们的应用,这些应用为氧化还原生物学的基本过程提供了新的启示。在几类次磺酸探针中,基于二甲酮的 C-亲核试剂具有优异的化学选择性和与串联 MS 的相容性。具有生物缀合手柄的细胞可渗透二甲酮衍生物能够检测活细胞中的 S- 苯基化。C-亲核试剂库的深入筛选产生了几个实体,它们的反应性比二甲双胍显着增强,同时保持选择性,以及能够实现受控靶标释放的可逆线性 C-亲核试剂。C-Nucleophiles 也已在标签转换方法中实施以检测 S-硫化。最近,
在可行的情况下,以位点为中心的定量蛋白质组学提供的丰富信息不应受到细胞裂解的氧化产物的污染。因此,现代氧化还原化学蛋白质组学方法需要在天然环境中发挥作用的具有低细胞毒性、良好细胞渗透性和竞争动力学的化学选择性探针。随着我们对硫化学和氧化还原信号的理解不断发展,新发现的微生物、植物、细胞、组织和疾病模型中的半胱氨酸 OxiPTM 应该会创新地促进机制和治疗研究。