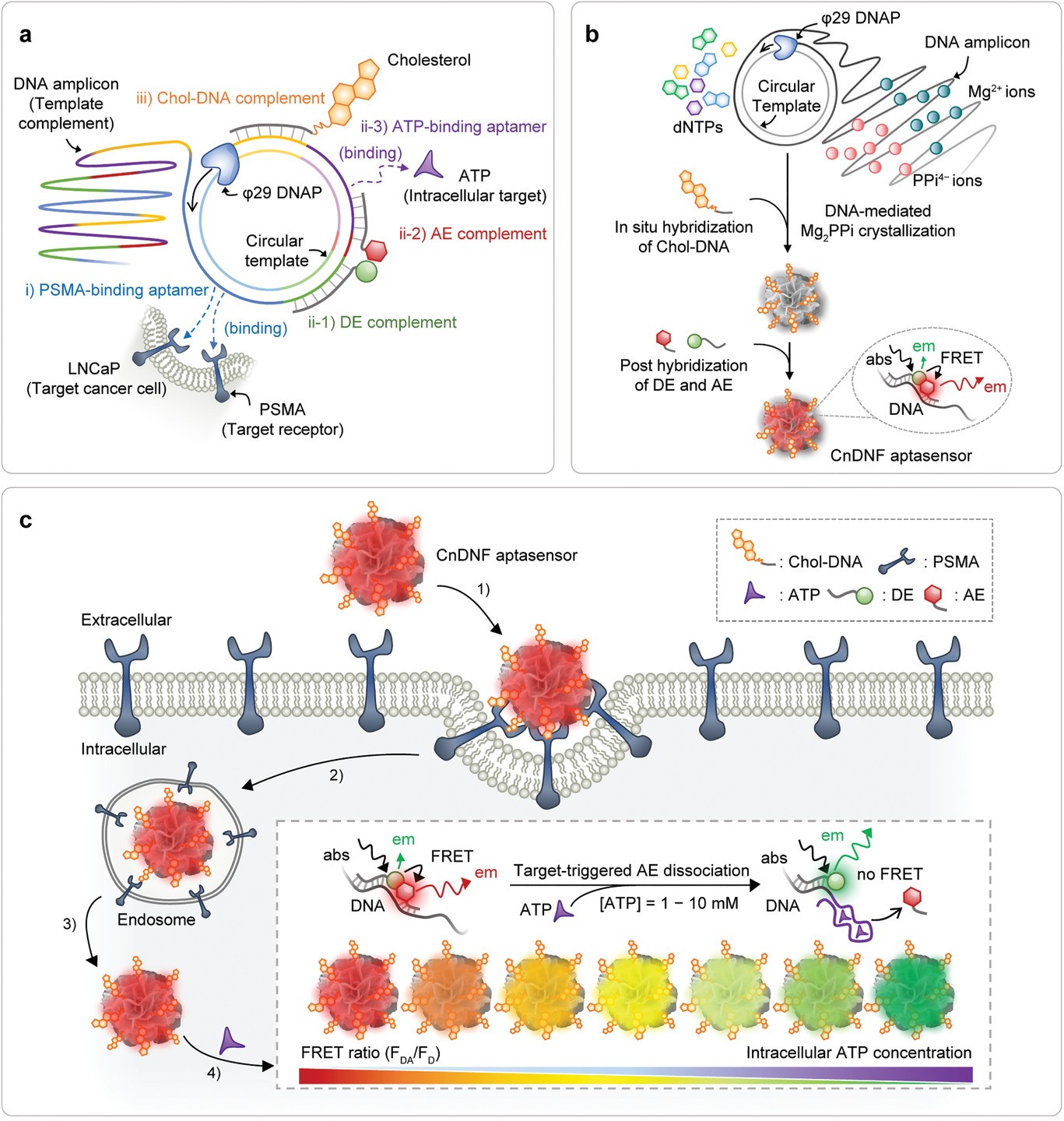
组会讲课人员:钱进
Tumor‐Targeting Cholesterol‐Decorated DNA Nanoflowers for Intracellular Ratiometric Aptasensing
肿瘤靶向型胆固醇修饰的DNA纳米花用于细胞内比率适配体传感
主讲人:钱进
Advanced Materials (IF 32.086) Pub Date: 2021-02-08, DOI: 10.1002/adma.202007738
Abstract:
Probing endogenous molecular profiles is of fundamental importance to understand cellular function and processes. Despite the promise of programmable nucleic‐acid‐based aptasensors across the breadth of biomolecular detection, target‐responsive aptasensors enabling intracellular detection are as of yet infrequently realized. Several challenges remain, including the difficulties in quantification/normalization of quencher‐based intensiometric signals, stability issues of the probe architecture, and complex sensor operations often necessitating extensive structural modeling. Here, the biomimetic crystallization‐empowered self‐assembly of a tumor‐targetable DNA–inorganic hybrid nanocomposite aptasensor is presented, which enables Förster resonance energy transfer (FRET)‐based quantitative interpretation of changes in the cellular target abundance. Leveraging the design programmability and high‐throughput fabrication of rolling circle amplification‐driven DNA nanoarchitecture, this designer platform offers a method to self‐assemble a robust nanosensor from a multifunctionality‐encoded template that includes a cell‐targeting aptamer, a ratiometric aptasensor, and a cholesterol‐decorating element. Taking prostate cancer cells and intracellular adenosine triphosphate molecules as a model system, a synergistic effect in the targeted delivery by cholesterol and aptamers, and the feasibility of quantitative intracellular aptasensing are demonstrated. It is envisioned that this approach provides a highly generalizable strategy across wide‐ranging target systems toward a biologically deliverable nanosensor that enables quantitative monitoring of the abundance of endogenous biomolecules.
摘要:
探测内源性分子谱对于了解细胞功能和过程具有根本的重要性。尽管基于核酸的可编程无源传感器在生物分子检测的广度上大有可为,但能够进行细胞内检测的目标反应无源传感器至今仍未实现。一些挑战依然存在,包括基于淬火剂的强化信号的量化/标准化困难,探针结构的稳定性问题,以及复杂的传感器操作往往需要大量的结构建模。在此,我们提出了一种生物仿生结晶技术,即自组装的肿瘤靶向DNA-无机混合纳米复合适体传感器,它可以实现基于福斯特共振能量转移(FRET)的对细胞靶标丰度变化的定量解释。利用滚动圈放大驱动的DNA纳米结构的设计可编程性和高通量制造,这个设计者平台提供了一种方法,从多功能编码的模板中自我组装一个强大的纳米传感器,其中包括一个细胞靶向的适配体,一个比率测量的适配体和一个胆固醇装饰元件。以前列腺癌细胞和细胞内三磷酸腺苷分子为模型系统,证明了胆固醇和诱导剂定向传递的协同效应,以及细胞内定量诱导的可行性。据设想,这种方法提供了一个高度通用的战略,跨越广泛的目标系统,实现了生物传递的纳米传感器,能够定量监测内源生物分子的丰度。